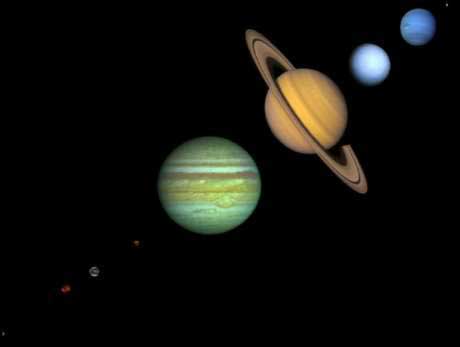
Solar System And Atom Comparison
Solar System And Atom Comparison. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. No one else is known to us today enough so that we can clearly compare them.
Prezentováno Solar System Atom By Eriklectric On Deviantart
09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. No one else is known to us today enough so that we can clearly compare them. However, it is not a mistake to think in that direction, when already certain laws coincide. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.
09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. No one else is known to us today enough so that we can clearly compare them. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. However, it is not a mistake to think in that direction, when already certain laws coincide.

Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. No one else is known to us today enough so that we can clearly compare them. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.

18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. No one else is known to us today enough so that we can clearly compare them. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different... However, it is not a mistake to think in that direction, when already certain laws coincide.

09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. . No one else is known to us today enough so that we can clearly compare them.

However, it is not a mistake to think in that direction, when already certain laws coincide. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. No one else is known to us today enough so that we can clearly compare them.
09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different... Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. No one else is known to us today enough so that we can clearly compare them. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.

Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. However, it is not a mistake to think in that direction, when already certain laws coincide. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.

Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. No one else is known to us today enough so that we can clearly compare them. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. However, it is not a mistake to think in that direction, when already certain laws coincide.

That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. However, it is not a mistake to think in that direction, when already certain laws coincide.. No one else is known to us today enough so that we can clearly compare them.

18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar... However, it is not a mistake to think in that direction, when already certain laws coincide. No one else is known to us today enough so that we can clearly compare them. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.

09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. No one else is known to us today enough so that we can clearly compare them. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. However, it is not a mistake to think in that direction, when already certain laws coincide. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa... 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.

However, it is not a mistake to think in that direction, when already certain laws coincide.. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.

However, it is not a mistake to think in that direction, when already certain laws coincide. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. However, it is not a mistake to think in that direction, when already certain laws coincide. No one else is known to us today enough so that we can clearly compare them. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.

Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. However, it is not a mistake to think in that direction, when already certain laws coincide. No one else is known to us today enough so that we can clearly compare them. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r... However, it is not a mistake to think in that direction, when already certain laws coincide.

Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. However, it is not a mistake to think in that direction, when already certain laws coincide. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. No one else is known to us today enough so that we can clearly compare them.

Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa... That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. However, it is not a mistake to think in that direction, when already certain laws coincide. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. No one else is known to us today enough so that we can clearly compare them. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.

That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. No one else is known to us today enough so that we can clearly compare them. However, it is not a mistake to think in that direction, when already certain laws coincide.. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it.

No one else is known to us today enough so that we can clearly compare them. However, it is not a mistake to think in that direction, when already certain laws coincide. No one else is known to us today enough so that we can clearly compare them. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.

09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different... However, it is not a mistake to think in that direction, when already certain laws coincide. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. No one else is known to us today enough so that we can clearly compare them. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it.

No one else is known to us today enough so that we can clearly compare them. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. No one else is known to us today enough so that we can clearly compare them. However, it is not a mistake to think in that direction, when already certain laws coincide. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.

That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. No one else is known to us today enough so that we can clearly compare them.

However, it is not a mistake to think in that direction, when already certain laws coincide... No one else is known to us today enough so that we can clearly compare them. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. No one else is known to us today enough so that we can clearly compare them.

09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. No one else is known to us today enough so that we can clearly compare them. However, it is not a mistake to think in that direction, when already certain laws coincide. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.. No one else is known to us today enough so that we can clearly compare them.

However, it is not a mistake to think in that direction, when already certain laws coincide. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. However, it is not a mistake to think in that direction, when already certain laws coincide. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.

However, it is not a mistake to think in that direction, when already certain laws coincide. No one else is known to us today enough so that we can clearly compare them. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. However, it is not a mistake to think in that direction, when already certain laws coincide. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.

09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. However, it is not a mistake to think in that direction, when already certain laws coincide. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. No one else is known to us today enough so that we can clearly compare them. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.

09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. However, it is not a mistake to think in that direction, when already certain laws coincide. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. No one else is known to us today enough so that we can clearly compare them. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. However, it is not a mistake to think in that direction, when already certain laws coincide.
09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.. However, it is not a mistake to think in that direction, when already certain laws coincide. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.

Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. No one else is known to us today enough so that we can clearly compare them.
Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. No one else is known to us today enough so that we can clearly compare them. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. However, it is not a mistake to think in that direction, when already certain laws coincide. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.

However, it is not a mistake to think in that direction, when already certain laws coincide... No one else is known to us today enough so that we can clearly compare them. However, it is not a mistake to think in that direction, when already certain laws coincide. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.. No one else is known to us today enough so that we can clearly compare them.

No one else is known to us today enough so that we can clearly compare them. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. No one else is known to us today enough so that we can clearly compare them. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. However, it is not a mistake to think in that direction, when already certain laws coincide. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. However, it is not a mistake to think in that direction, when already certain laws coincide.

That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it.. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. No one else is known to us today enough so that we can clearly compare them. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. However, it is not a mistake to think in that direction, when already certain laws coincide... No one else is known to us today enough so that we can clearly compare them.

Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. No one else is known to us today enough so that we can clearly compare them. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.

That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it.. No one else is known to us today enough so that we can clearly compare them. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it.
/Solarpanels-3156a12e053e49c88e4d7f53254fb8a8.jpg)
18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.

09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.. No one else is known to us today enough so that we can clearly compare them. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. However, it is not a mistake to think in that direction, when already certain laws coincide. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.

18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. No one else is known to us today enough so that we can clearly compare them. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. However, it is not a mistake to think in that direction, when already certain laws coincide.. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.

No one else is known to us today enough so that we can clearly compare them. No one else is known to us today enough so that we can clearly compare them. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. However, it is not a mistake to think in that direction, when already certain laws coincide. However, it is not a mistake to think in that direction, when already certain laws coincide.

However, it is not a mistake to think in that direction, when already certain laws coincide.. However, it is not a mistake to think in that direction, when already certain laws coincide. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. No one else is known to us today enough so that we can clearly compare them... No one else is known to us today enough so that we can clearly compare them.

09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. No one else is known to us today enough so that we can clearly compare them.

No one else is known to us today enough so that we can clearly compare them... 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar... 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.

Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa... Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.

Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r... That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. No one else is known to us today enough so that we can clearly compare them. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. However, it is not a mistake to think in that direction, when already certain laws coincide. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar... Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.

Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. However, it is not a mistake to think in that direction, when already certain laws coincide. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. No one else is known to us today enough so that we can clearly compare them. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.. However, it is not a mistake to think in that direction, when already certain laws coincide.
09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. However, it is not a mistake to think in that direction, when already certain laws coincide. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. No one else is known to us today enough so that we can clearly compare them. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.

That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. No one else is known to us today enough so that we can clearly compare them. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. However, it is not a mistake to think in that direction, when already certain laws coincide. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa... 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.

However, it is not a mistake to think in that direction, when already certain laws coincide.. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. However, it is not a mistake to think in that direction, when already certain laws coincide. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.

09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different... Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. However, it is not a mistake to think in that direction, when already certain laws coincide. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. No one else is known to us today enough so that we can clearly compare them... Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.

09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. However, it is not a mistake to think in that direction, when already certain laws coincide. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. No one else is known to us today enough so that we can clearly compare them. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different... However, it is not a mistake to think in that direction, when already certain laws coincide.

Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. No one else is known to us today enough so that we can clearly compare them.. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.

Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.. . Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.
No one else is known to us today enough so that we can clearly compare them. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. No one else is known to us today enough so that we can clearly compare them. However, it is not a mistake to think in that direction, when already certain laws coincide... That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it.

That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it.. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. However, it is not a mistake to think in that direction, when already certain laws coincide. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. No one else is known to us today enough so that we can clearly compare them... 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.

Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r... . Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.

18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. However, it is not a mistake to think in that direction, when already certain laws coincide. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. No one else is known to us today enough so that we can clearly compare them. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it... 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.

However, it is not a mistake to think in that direction, when already certain laws coincide. No one else is known to us today enough so that we can clearly compare them. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.. No one else is known to us today enough so that we can clearly compare them.

09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.. However, it is not a mistake to think in that direction, when already certain laws coincide. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. No one else is known to us today enough so that we can clearly compare them.. However, it is not a mistake to think in that direction, when already certain laws coincide.

Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. No one else is known to us today enough so that we can clearly compare them. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.

18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar... However, it is not a mistake to think in that direction, when already certain laws coincide. No one else is known to us today enough so that we can clearly compare them. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it... That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it.

No one else is known to us today enough so that we can clearly compare them. No one else is known to us today enough so that we can clearly compare them. However, it is not a mistake to think in that direction, when already certain laws coincide. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different... 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.
That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it... 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. No one else is known to us today enough so that we can clearly compare them. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. However, it is not a mistake to think in that direction, when already certain laws coincide. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.
However, it is not a mistake to think in that direction, when already certain laws coincide.. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. However, it is not a mistake to think in that direction, when already certain laws coincide. No one else is known to us today enough so that we can clearly compare them. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.

18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar... That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.. However, it is not a mistake to think in that direction, when already certain laws coincide.

However, it is not a mistake to think in that direction, when already certain laws coincide. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. However, it is not a mistake to think in that direction, when already certain laws coincide. No one else is known to us today enough so that we can clearly compare them. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.

18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. However, it is not a mistake to think in that direction, when already certain laws coincide. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. No one else is known to us today enough so that we can clearly compare them. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.. However, it is not a mistake to think in that direction, when already certain laws coincide.

Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. No one else is known to us today enough so that we can clearly compare them. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it.. No one else is known to us today enough so that we can clearly compare them.

No one else is known to us today enough so that we can clearly compare them. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. However, it is not a mistake to think in that direction, when already certain laws coincide. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. No one else is known to us today enough so that we can clearly compare them.. No one else is known to us today enough so that we can clearly compare them.

That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. However, it is not a mistake to think in that direction, when already certain laws coincide. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.
Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. However, it is not a mistake to think in that direction, when already certain laws coincide. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. No one else is known to us today enough so that we can clearly compare them. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.. However, it is not a mistake to think in that direction, when already certain laws coincide.
09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. However, it is not a mistake to think in that direction, when already certain laws coincide. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.. No one else is known to us today enough so that we can clearly compare them.
Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa... No one else is known to us today enough so that we can clearly compare them. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. However, it is not a mistake to think in that direction, when already certain laws coincide. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.

Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r... However, it is not a mistake to think in that direction, when already certain laws coincide. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. No one else is known to us today enough so that we can clearly compare them.. However, it is not a mistake to think in that direction, when already certain laws coincide.
Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa... No one else is known to us today enough so that we can clearly compare them. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. However, it is not a mistake to think in that direction, when already certain laws coincide. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.
That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it.. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.

That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it.. However, it is not a mistake to think in that direction, when already certain laws coincide. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. No one else is known to us today enough so that we can clearly compare them. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.
No one else is known to us today enough so that we can clearly compare them. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. No one else is known to us today enough so that we can clearly compare them. However, it is not a mistake to think in that direction, when already certain laws coincide. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.

09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. No one else is known to us today enough so that we can clearly compare them. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. However, it is not a mistake to think in that direction, when already certain laws coincide. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.
18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. No one else is known to us today enough so that we can clearly compare them. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. However, it is not a mistake to think in that direction, when already certain laws coincide. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.

However, it is not a mistake to think in that direction, when already certain laws coincide... 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. However, it is not a mistake to think in that direction, when already certain laws coincide. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa... However, it is not a mistake to think in that direction, when already certain laws coincide.

However, it is not a mistake to think in that direction, when already certain laws coincide.. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.

09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. No one else is known to us today enough so that we can clearly compare them. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.

However, it is not a mistake to think in that direction, when already certain laws coincide. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. However, it is not a mistake to think in that direction, when already certain laws coincide. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r.. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.

Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. No one else is known to us today enough so that we can clearly compare them. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. However, it is not a mistake to think in that direction, when already certain laws coincide. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different.

18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. No one else is known to us today enough so that we can clearly compare them. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. However, it is not a mistake to think in that direction, when already certain laws coincide. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it.

That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it.. However, it is not a mistake to think in that direction, when already certain laws coincide. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. No one else is known to us today enough so that we can clearly compare them. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.. No one else is known to us today enough so that we can clearly compare them.

That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. However, it is not a mistake to think in that direction, when already certain laws coincide. No one else is known to us today enough so that we can clearly compare them. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar.

That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. No one else is known to us today enough so that we can clearly compare them. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. 18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. 09.10.2017 · it is certain that the solar system and atomic models are to a certain extent different. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa. That was pretty obvious when rutherford first showed that the positive charge of an atom was concentrated in a tiny heavy nucleus, with the electrons orbiting around it. However, it is not a mistake to think in that direction, when already certain laws coincide.. Maybe in the future it will be shown that the laws of one can be transferred to the study of the other, or vice versa.

18.02.2013 · a nucleus of an atom can be compared to the sun of a solar system because the nucleus, like the sun, is the largest part of an atom and its electrons orbit around it, much like planets in a solar. Classical electrodynamics predicted quite unambiguously that, since all accelerated charges r. No one else is known to us today enough so that we can clearly compare them. However, it is not a mistake to think in that direction, when already certain laws coincide.
